As global population aging intensifies, extending “health span” and precisely intervening in “biological age” have become core biomedical challenges. Recently, an interdisciplinary team of experts from Southern University of Science and Technology (SUSTech), University of Electronic Science and Technology of China, Macau University of Science and Technology (MUST), the Max Planck Institute for Evolutionary Biology in Germany, the Leibniz Institute on Aging, and other top research institutions — with Professor Zhu Jian-Kang (MUST) as a corresponding author — published a comprehensive review in the premier aging research journal Ageing Research Reviews (IF=13, JCR Q1). The article systematically explores the use of short-lived model organisms such as C. elegans, fruit flies, and African killifish, combined with automated high-throughput screening (HTS) and artificial intelligence (AI), to accelerate breakthroughs, address technical challenges, and chart clinical translation pathways for novel geroprotectors.

Figure 1:Article title: “High-Throughput Screening for Ageing and Age-Related Disease Drug Discovery: Advances and Challenges”. Corresponding authors are Prof. Jian-Kang Zhu, Prof. Alessandro Cellerino, Dr Judith Bossen, Prof. Bo Xian and Prof. Yu-Xuan Lyu.
Aging is a core driver of cancers, cardiovascular diseases, and neurodegenerative disorders. Although modern medicine has significantly extended average human lifespan, health span has not kept pace, leaving many older adults in states of disability and multimorbidity.
In drug discovery pipelines, traditional cell lines cannot model complex organismal aging. While vertebrate models like zebrafish and mice are informative, their multi-year lifespans entail high costs and long R&D cycles. This review focuses on short-lived models that share highly conserved aging mechanisms with humans — C. elegans and fruit flies — and highlights the emerging vertebrate model African killifish as a key bridge in translational medicine. It proposes a new aging-drug discovery paradigm that integrates multi-omics, automated monitoring, and AI-driven visual reasoning.
1. Three Model Organisms: Pioneers for Anti-Aging Drug Screening
To bridge the gap between in vitro assays and human clinical applications, researchers are building tiered screening platforms using organisms of increasing biological complexity:
C. elegans: With a 15–25 day lifespan and fully transparent body, it is the classic model for studying insulin/IGF-1 signaling, mitochondrial stress, and proteostasis imbalance.
Drosophila melanogaster: Shares ~75% homology with human disease-related genes. Its 1–2 month lifespan, clear sex differences, and circadian rhythms make it ideal for studying CNS, gut, and muscle aging as well as sex-specific interventions.
African killifish (Nothobranchius furzeri): The shortest-lived vertebrate in the lab (4–6 months), perfectly filling the gap between invertebrates and mammals. It possesses adaptive immunity and complex tissue regeneration capabilities characteristic of vertebrates.
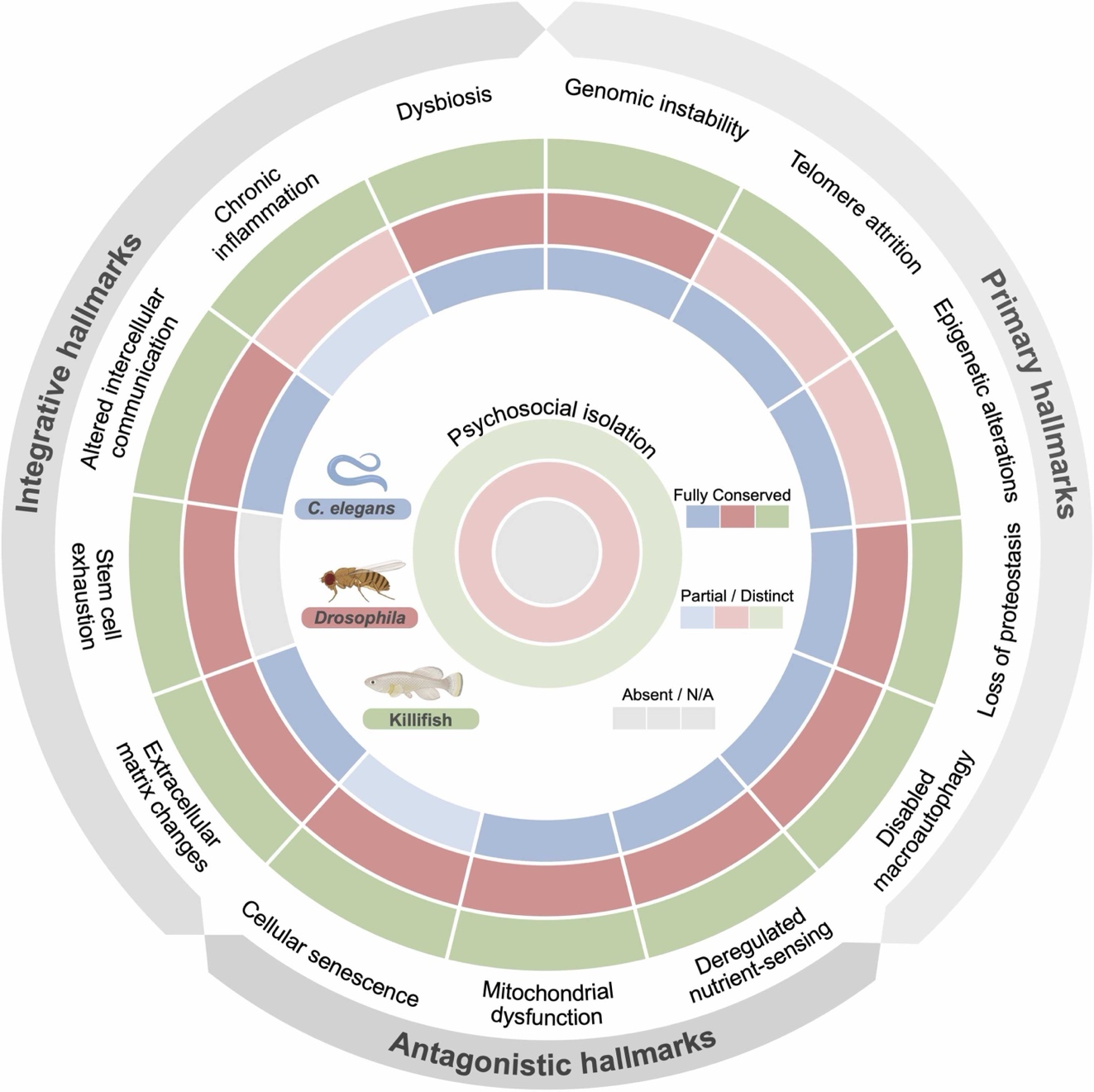
Figure 2:These three model organisms: C. elegans (worms), Drosophila (fruit flies), and the African turquoise killifish, recapitulate fundamental ageing mechanisms, including the majority of hallmarks, facilitating the study of conserved pathways relevant to human ageing and geroprotector discovery.
2. From Manual Counting to Digital-Intelligent Monitoring
Traditional aging research relies heavily on manual observation — low throughput and subjective. The review details cutting-edge hardware platforms such as WormBot, the Lifespan Machine, and DIAMonDS (Drosophila Individual Activity Monitoring and Detection System). These platforms integrate high-throughput video tracking, microfluidic chips, and infrared sensors to automate lifelong monitoring of thousands of individuals from birth to death. This “unattended” mode greatly improves throughput while ensuring objectivity and reproducibility.
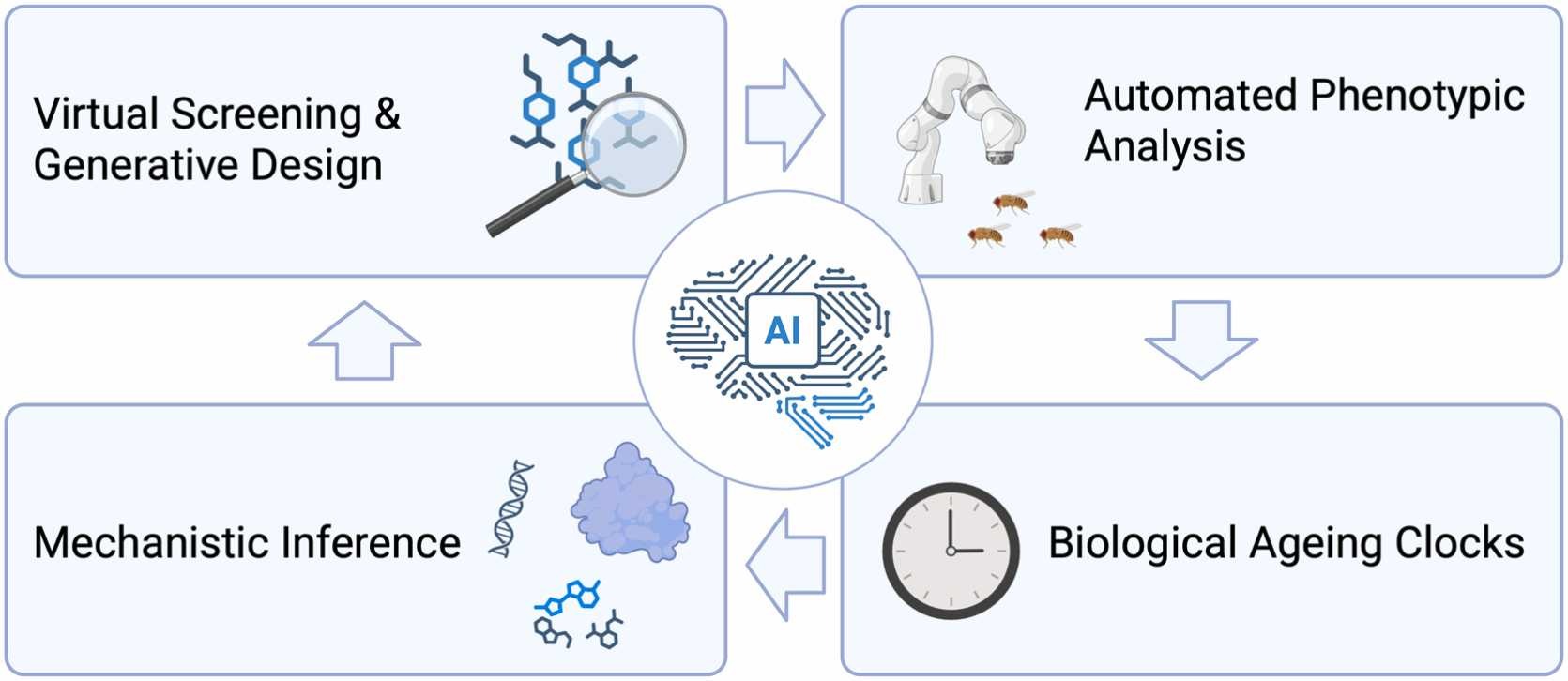
Figure 3:AI enhances every stage of high-throughput screening (HTS) in ageing research and drug discovery by enabling: virtual and generative compound design; automated phenotypic screening using computer vision; biological ageing clocks and mechanistic inference, facilitating target discovery and mode-of-action elucidation.
3. Targeting Aging Hallmarks: From Living Longer to Aging Slower
The review emphasizes that modern screening goals have shifted from merely extending lifespan to deeply intervening in the hallmarks of aging:
Primary hallmarks: Using fluorescent reporter systems to monitor proteostasis imbalance and oxidative stress in real time.
Antagonistic hallmarks: Dynamically tracking mitochondrial morphology (from networked to fragmented) to screen metabolic regulators that precisely improve mitochondrial function.
Integrative hallmarks:
"Smurf" assay: Monitoring systemic “blue leakage” due to loss of gut integrity predicts mortality risk in advance — a key indicator of gut barrier dysfunction and systemic inflammation.
Behavioral tracking: Networked automated feeding systems (e.g., from Stanford) not only precisely control calorie intake but also perform visual associative learning tasks, evaluating drug effects on vertebrate cognition and social behavior.
4. The Reverse Translation Strategy: Accelerating Drug Repurposing
Unlike the traditional linear “bench-to-bedside” logic, the review highlights the great potential of “reverse translation.” Taking metformin, SGLT2 inhibitors, and GLP-1 receptor agonists as examples: these drugs were originally developed for metabolic diseases, but large-scale epidemiological data revealed their significant anti-aging potential. By rapidly performing high-throughput mechanistic validation in short-lived models, researchers can quickly determine whether these drugs act through conserved longevity pathways (e.g., AMPK or mTOR), dramatically shortening the cycle from clinical observation to mechanism confirmation to broader applications.
Future aging drug discovery will be a “dry–wet” hybrid pipeline: from AI-assisted virtual design of billions of molecules, to automated robotic bio-validation, to organ-on-a-chip human simulation. This closed-loop system is compressing R&D timelines at unprecedented speed, helping humanity cross the “healthspan gap” and advance at full speed toward the goal of healthy aging.
The co-first authors of this paper are Xingkun Ji (PhD student) and JiajunLei (Master’s student) from the School of Medicine / Institute of Advanced Biotechnology at SUSTech, and Yan Pan (undergraduate student) from the School of Medicine, University of Electronic Science and Technology of China. Corresponding authors include Professor Yu-Xuan Lyu (SUSTech), Professor Bo Xian (UESTC), Professor Jian-Kang Zhu (MUST), Professor Alessandro Cellerino (Leibniz Institute on Aging / University of Pisa), and Dr. Judith Bossen (Max Planck Institute for Evolutionary Biology / Kiel University). The study also received support from Professor Fangyi Chen (SUSTech College of Engineering), Professor Jiongming Lu (Shanghai Institute of Nutrition and Health, CAS), and several other research institutions in China and abroad.




























